 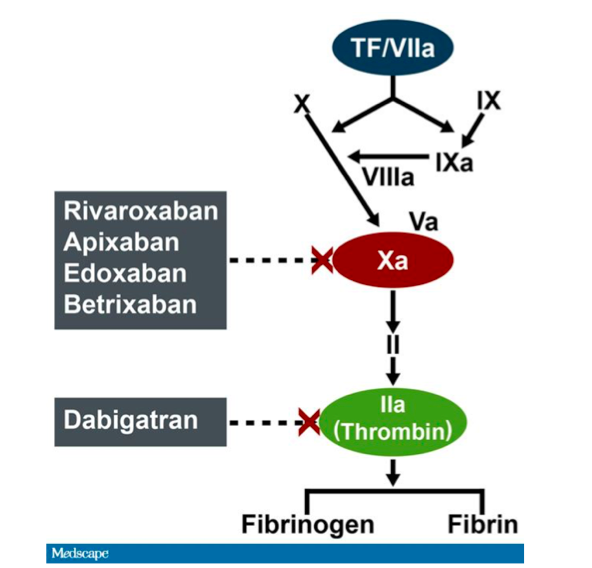
Dabigatran etexilate (aDabiFab) is the only direct thrombin inhibitor among the authorized NOACs and was the first one to be demonstrated more effective and safer than warfarin for AF 3 and has a similar efficacy and safety for VTE patients. In 2009 the introduction of non-vitamin K antagonist oral anticoagulants (NOACs) in the market as an effective and safer alternative for the prevention of thromboembolic events in atrial fibrillation (AF) patients 1 and the treatment and prevention of recurrences in patients with venous thromboembolic events (VTE) 2 drastically changed the current pharmacopeia and routine clinical treatment of these two conditions. Keywords: anticoagulant drugs, non-vitamin K antagonist oral anticoagulants, reversal agents, idarucizumab, major bleeding Furthermore, we review the current recommendations and experts’ point of view about the use of antidotes/reversal agents in patients reporting a major bleeding event. This review briefly summarizes the experimental evidence about effectiveness and safety of idarucizumab. Idarucizumab was approved in 2015 by the US Food and Drugs Administration and European Medicines Agency for reversal of anticoagulation activity in dabigatran-treated patients. Idarucizumab is a humanized monoclonal antibody fragment that binds specifically to dabigatran. Despite that, a strong need for antidotes/reversal agents is still reported by several physicians. 
1Department of Neuroscience, IRCCS – Istituto di Ricerche Farmacologiche “Mario Negri”, Milan, Italy 2Institute of Cardiovascular Sciences, University of Birmingham, Birmingham, UK 3Department of Internal Medicine and Medical Specialties, Sapienza-University of Rome, Rome, Italy 4Department of Biomedical, Metabolic and Neural Sciences, University of Modena and Reggio Emilia, Policlinico di Modena, Modena, ItalyĪbstract: Use of non-vitamin K antagonist oral anticoagulants is spreading in the real world.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
